THE 1723 CATALOGUE
A1
A2
A3
A4
A5
A6
A7
A8
A9
A10
A11
A12
A13
A14
A15
A16
A17
A18
A19
A20
A21
A22
A23
A24
A25
A26
A27
A28
A29
A30
A31
A32
A33
A34
A35
A36
A37
A38
A39
A40
A41
A42
A43
A44
A45
A46
A47
A48
A49
A50
A51
A52
A53
A54
A55
A56
A57
A58
A59
A60
A61
A62
A63
A64
A65
A66
A67
A68
A69
A70
A71
A72
A73
A74
A75
A76
A77
A78
A79
A80
A81
A82
A83
A84
A85
A86
A87
A88
A89
A90
A91
A92
A93
A94
A95
A96
A97
A98
A99
A100
A101
A102

A103
A104
A105
A106
A107
A108
A109
A110
A111
A112
A113
A114
A115
A116
A117
A118
A119
A120
A121
A122
A123
A124

1 Alfred Claghorn Potter, The Library of Harvard University: Descriptive and Historical Notes, 3rd ed. (Cambridge, Mass., 1915), pp. 13–15, provides a concise description of the origins and early development of the library, and lists its benefactors.
2 Samuel Eliot Morison, The Founding of Harvard College (Cambridge, Mass., 1935), pp. 263–70, especially pp. 264–66, describes John Harvard’s legacy; see also his Harvard College in the Seventeenth Century (Cambridge, Mass, 1936), I, 285–97; the inventory of John Harvard’s books is reprinted in Harvard College Records, PCSM, 15–16 (1925) (hereafter cited as “Records”), 158–66.
3 Morison, Founding, pp. 272–89, gives an account of the Old College building, which was begun in 1638 but not completed until four years after the library was installed in it; a conjectural floor plan, drawn by H. R. Shurtleff on the basis of existing documents, faces p. 284. Kenneth E. Carpenter, The First 350 Years of the Harvard University Library: Description of an Exhibition (Cambridge, Mass., 1986), p. 5, reprints this plan, with a rendering of the supposed interior by F. W. Hartwell; but the fenestration of the rendering does not agree with the plan, and there is no evidence for the low lectern-style shelving and benches, typical of European libraries of a much earlier period. The Old College was poorly constructed and much in need of repairs as early as 1651; Cotton Mather noted that its unoccupied ruins still stood in 1695.
4 Shurtleff’s floor plans of Harvard Hall are printed by Morison, 17th Century, facing p. 429; Carpenter, First 350 Years, p. 6, reprints the plan of the second floor.
5 Records, pp. lxxxvii, note 1, 350 and 712.
6 He was a wealthy London Baptist (d. 1731) who never visted America but gave his support to Harvard College as the leading nonconformist foundation in the colonies. He gave the College’s first two endowed chairs, the Hollis Professorships of Divinity (1723) and of Mathematics and Natural Philosophy (1727), sent books to the library, and encouraged others to do likewise. He was the great-uncle of Thomas Hollis of Lincoln’s Inn.
7 Documents from the Harvard University Archive, 1638–1750, ed. R.W. Lovett, PCSM, 49–50 (1975) (hereafter cited as “Documents”), 500–501.
8 See the illustrations in John Willis Clark, The Care of Books (Cambridge, 1901), figs. 110–12.
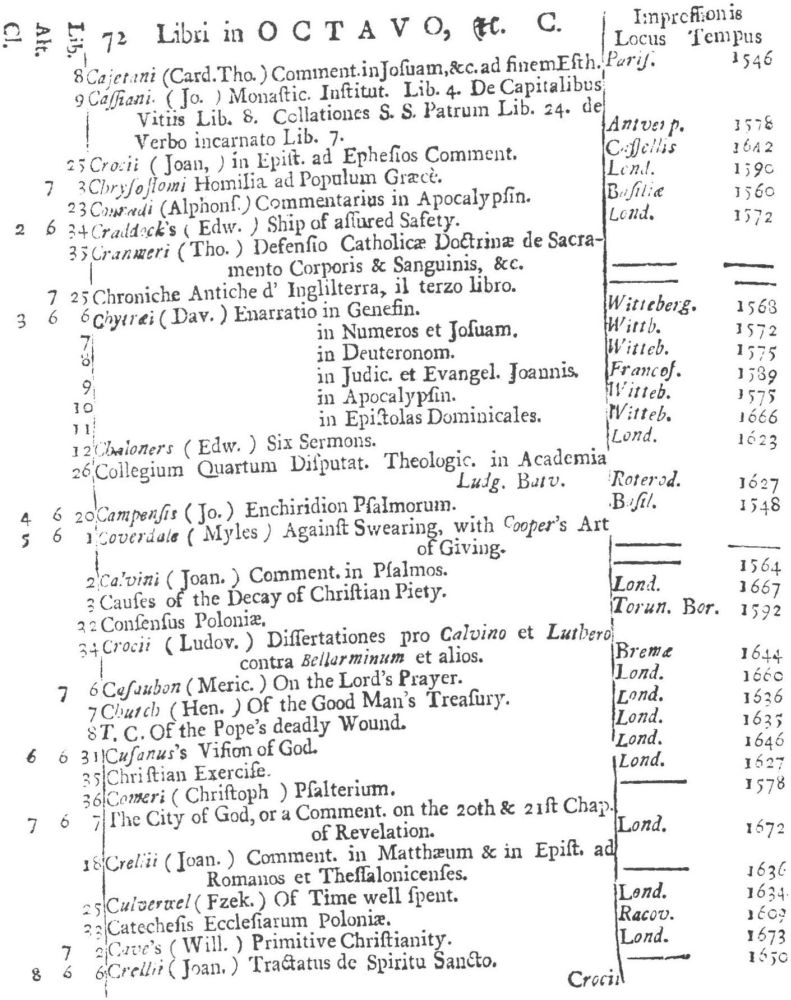
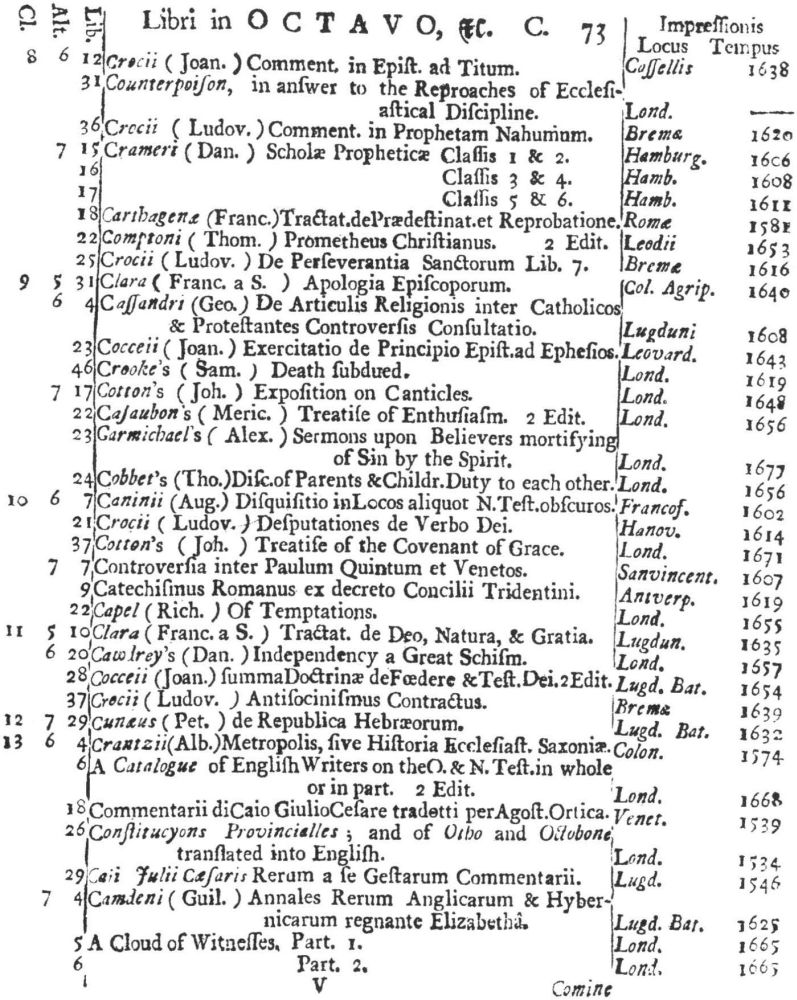
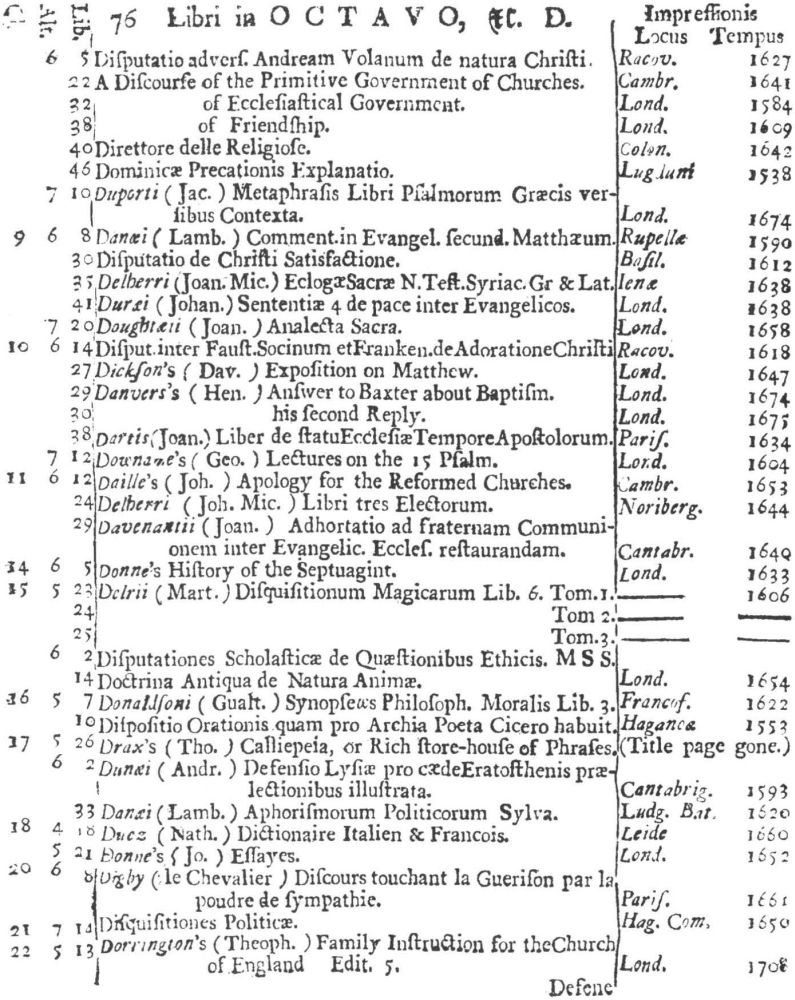
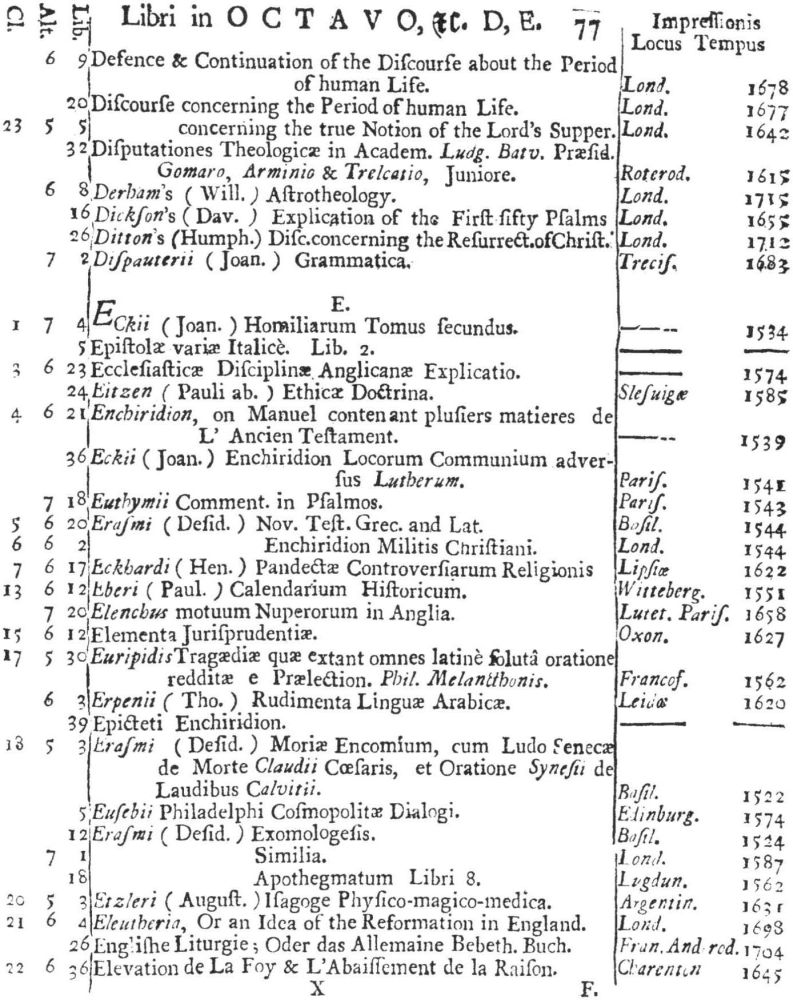
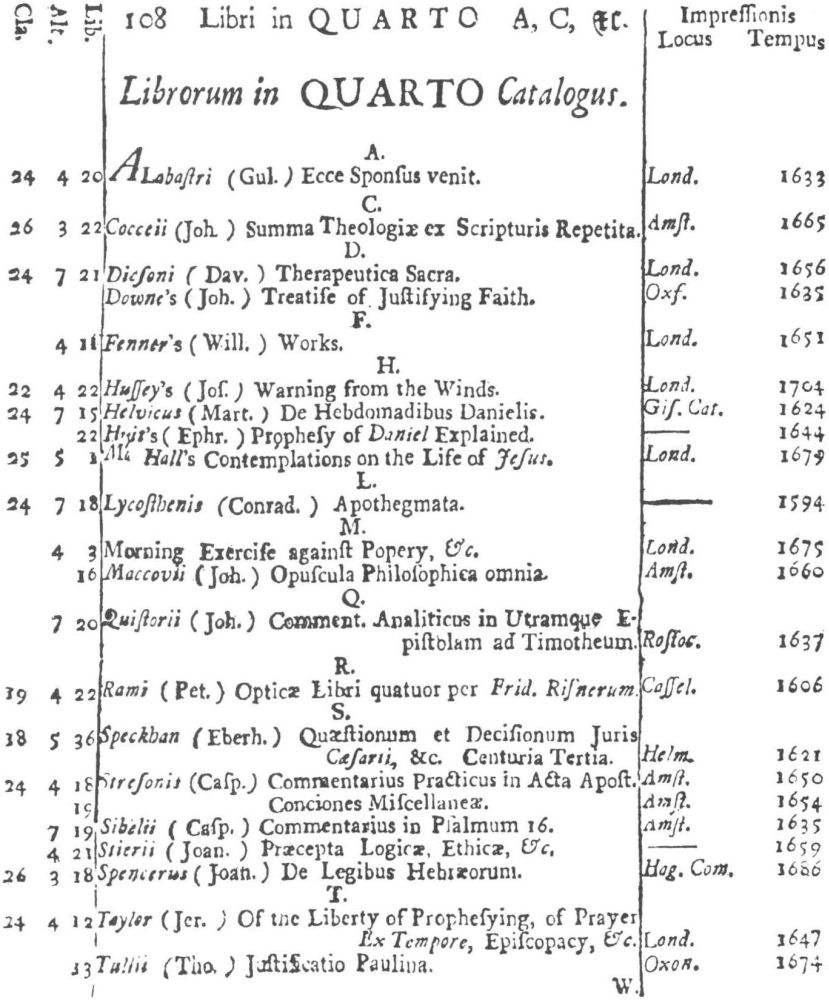
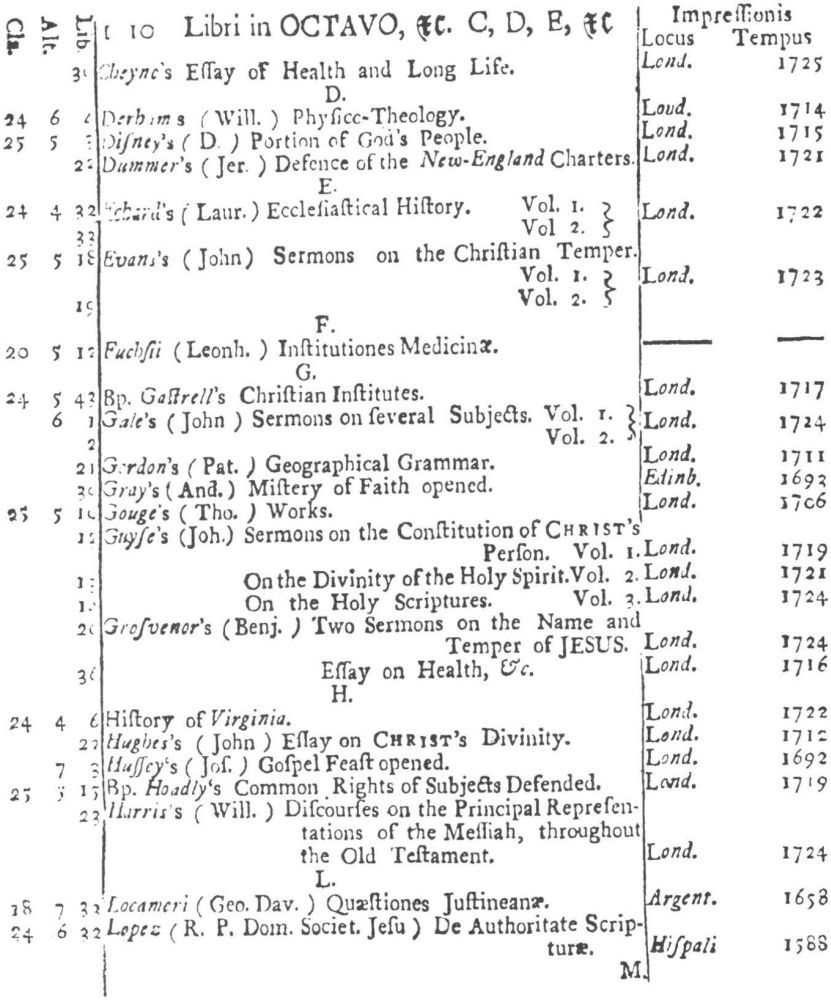
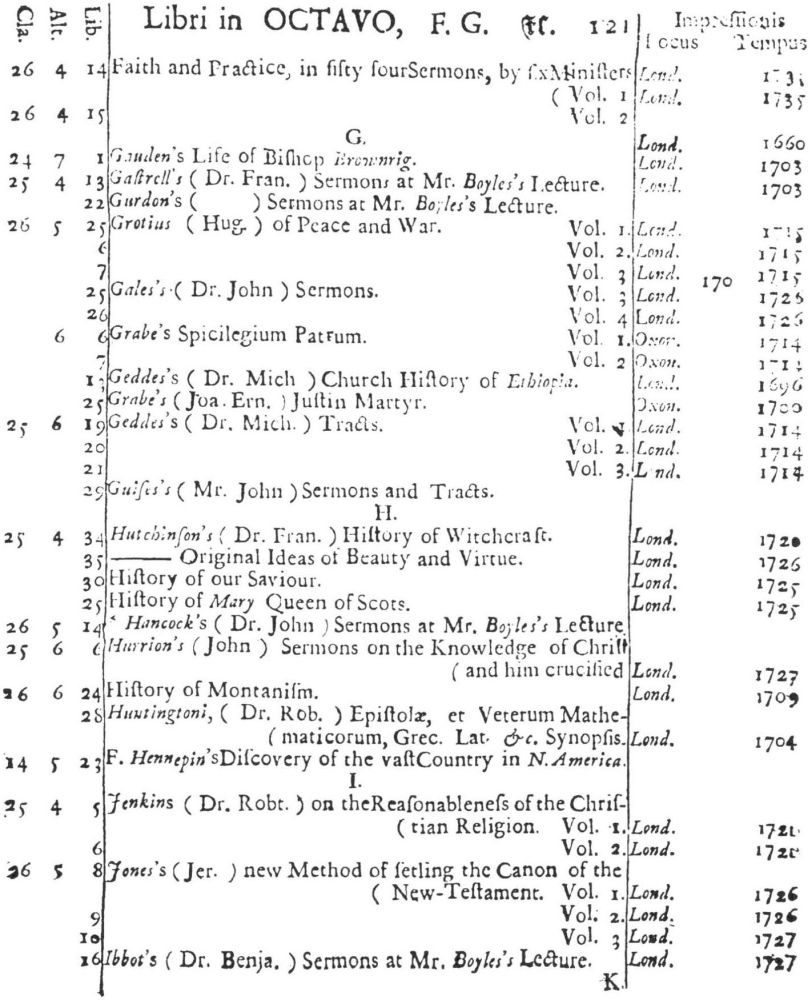
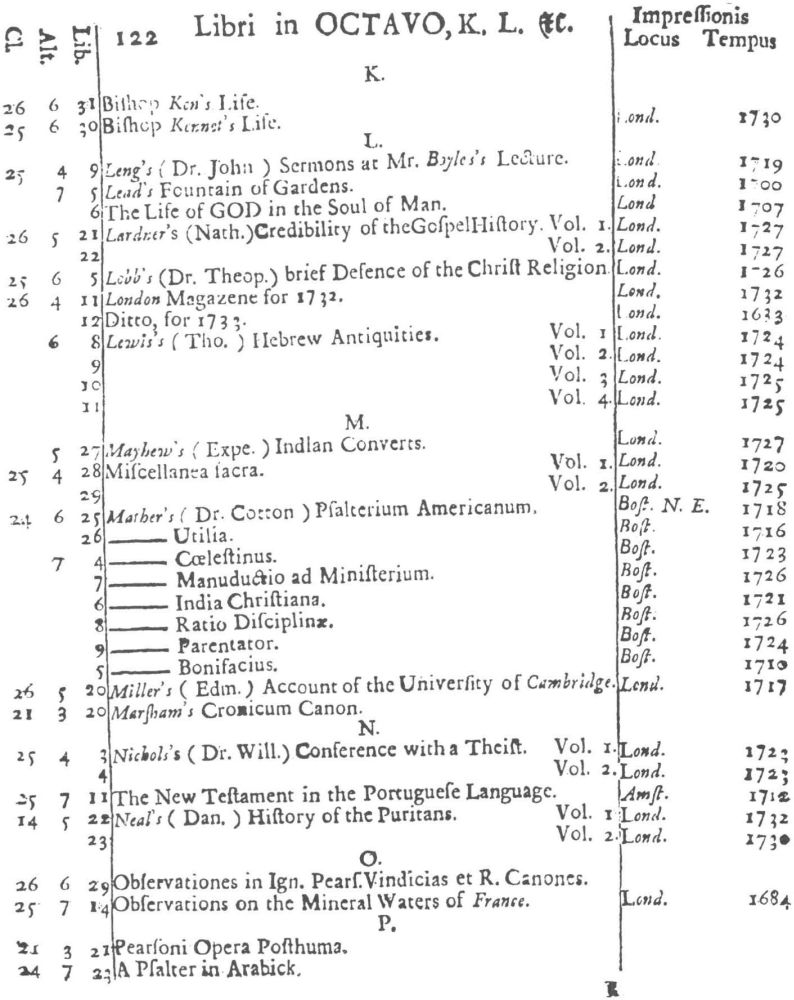
9 Clark, Care of Books, p. 243: “The word ‘class’ (classis) is used at the University Library, Cambridge, in 1584, instead of the ancient ‘stall’, and afterwards superseded it entirely. For instance, when a Syndicate was appointed in 1713 to provide accommodation for Bishop Moore’s Library, the bookcases are described as Thecæ sive quas vocant classes.” The “Præmonitio” of the 1723 Catalogus calls the cases plutei and adds “quos Classes vocamus,” and the shelf-marks as printed invariably refer to the “Cl[assis].”
10 See further below, p. XXVI. Hugh Amory suggests, on the basis of the entry of “Francisci Lamberti” as “Scilambertus, Fran.,” in the 1723 Catalogus (p. 92), that authors’ names were written in direct order in the compiler’s source.
11 Documents, p. 556.
12 For a facsimile of the broadside, and of the title page of the only book now surviving from John Harvard’s gift, see The Houghton Library, 1942–1967 (Cambridge, 1967), pp. 2–3.
13 Not fifty-eight, as stated in the broadside.
14 Potter, Library, pp. 122–24, lists the most substantial benefactors.
15 Carpenter, First 350 Years, p. 19, reproduces a plan of the new library room; the rendering of “The Library as it probably looked in 1790” on p. 18 is, however, fanciful.
16 First 350 Years, above, n. 3.
17 The Alcove Lists of about 1765, written in stitched folio pamphlets, are preserved in the Harvard University Archives (UA III.50.15.31.5); they are much worn, and interlined or otherwise revised, and clean and somewhat simplified copies were required a few years later, which also survive (UA III.50.15.40).
18 Thomas Hollis V died on January 1, 1774, and had ceased sending wholesale lots of books to Cambridge several years earlier; his friend and heir Thomas Brand Hollis continued the family benefactions (later shelved in Alcove 2.5, 4.5, and 6.5).
19 Thomas Jay Siegel, “Governance and Curriculum at Harvard College in the 18th Century,” Harvard Ph.D. Thesis, 1990, University Microfilms microfiche 90-35554 (1995).
20 Quoted in Potter, Library, p. 16.
21 UA III.50.15.39 (A, B, & C).
22 See the graph in Carpenter, First 350 Years, p. 199.
23 See Potter, Library, p. 169 for the roll of early 19th-century librarians.
24 I, xiii, note; an annotated copy in the Harvard University Archives has been helpful in identifying some elusive entries in the earlier catalogues.
25 Quoted in Clark, Care of Books, p. 195.
26 Daniel Neal, The History of New-England (London, 1720), pp. 183–84; apparently basing his account on Mather’s Magnalia.
27 A unique copy of the catalogue is in the Boston Public Library.
28 Samuel Eliot Morison, Harvard in the Seventeenth Century (Cambridge, Mass., 1936), p. 291.
29 Documents, p. 388.
30 Records, p. 195.
31 “Library Papers: Chronological series”, Harvard University Archives, UA III.50.27.14vt.
32 So in Government printing, the bills were made out to the bookseller, not to the printer. Rollo G. Silver, “Government Printing in Massachusetts Bay, 1700–1750,” PAAS, 68 (1958), [135]–62, at pp. 144–45.
33 Rollo G. Silver, “Publishing in Boston, 1726–1757,” PAAS, 66 (1956), 17–56, at p. 24.
34 Records, p. 503; the bill, from the “Library Papers”, is transcribed in Appendix 1, below, courtesy of the Harvard University Archives. Calculated in pounds, shillings and pence (£.s.d.): £1 = 20s.; 1s. = 12d.
35 Printed in Lawrence C. Wroth, The Colonial Printer, 2nd ed., rev. & enl. (Charlottesville, Va., 1964), p. 184; Wroth’s comparison of colonial and London charges unfortunately ignores such concealed costs.
36 Records, p. 699.
37 Records, p. 503.
38 Documents, p. 543; a copy of the letter is in the Massachusetts Historical Society, Misc. MSS.
39 Ibid., p. 558.
40 llustrated in Blickling Hall (London: National Trust, 1987), p. 55; I am grateful to David McKitterick for bringing this reference to my attention; interestingly, Harvard had hoped to acquire this library in 1741.
41 For the present location of these copies, I use the library symbols of the pre-1956 National Union Catalog.
42 Documents, pp. 500 and 560.
43 “Library Papers,” Harvard University Archives.
44 Records, p. 646.
45 Siegel, “Governance of Harvard,” p. 276.
46 Corporation Records, Harvard University Archives, UA 1.5.30.2, v. 2 (1750–78), pp. 395–95A.
47 Memoir of Hezekiah Packard D.D. (Brunswick [Me.], 1850), p. 18.
48 “Library Papers,” transcribed in Appendix 1, below, courtesy of the Harvard University Archives.
49 I am most grateful to Mr. Jerome Anderson for allowing me to examine this copy, in his collection.
50 Corporation Records, Harvard University Archives, v. 3, pp. 371–72.
51 Houghton Library, call no. X 16.98.
52 This manuscript is in the American Antiquarian Society Library.
53 Siegel, “Governance of Harvard,” p. 317.
54 Houghton Library, call no. *AC7.B8188.Zz711p: a collection of 10 plays by Susanna Centlivre and others, 1692–1711, with Browne’s note, “bound 1752”. The earliest student drama society dates from 1758.
55 Elizabeth C. Reilly, “The Wages of Piety: The Boston Book Trade of Jeremy Condy,” in Printing and Society in Early America, ed. W. L. Joyce et al. (Worcester, Mass., 1983), pp. [83]–131; see also Siegel, “Governance of Harvard”, pp. 313–15.
56 Cf. Samuel Eliot Morison, The Intellectual Life of Colonial New England (Ithaca, N.Y., 1956, repr. 1987), pp. 148–50.
57 A contemporary list of these is in the University Archives. Many of the books themselves were identified by John Langdon Sibley, and a shelflist (by the original shelf mark) of those in Houghton is maintained on fiches, known as the “Old Library” file. I am not aware of any systematic search for survivors outside Harvard, however.
58 James Gilreath, “American Book Distribution,” PAAS, 95 (1985), 524–25.